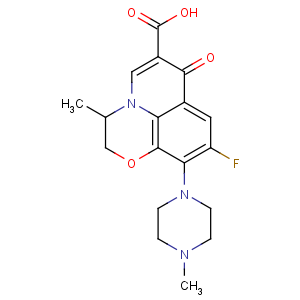
Title: Ofloxacin
CAS Registry Number: 82419-36-1
CAS Name: 9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7
H-pyrido[1,2,3-
de]-1,4-benzoxazine-6-carboxylic acid
Synonyms: ofloxacine
Manufacturers' Codes: DL-8280; HOE-280
Trademarks: Exocin (Allergan); Flobacin (Sigma-Tau); Floxil (Janssen-Cilag); Floxin (Ortho-McNeil); Monoflocet (Aventis); Ocuflox (Allergan); Oflocet (Aventis); Oflocin (GSK); Tarivid (Aventis)
Molecular Formula: C18H20FN3O4
Molecular Weight: 361.37
Percent Composition: C 59.83%, H 5.58%, F 5.26%, N 11.63%, O 17.71%
Literature References: Broad spectrum, fluorinated quinolone antibacterial. Prepn: I. Hayakawa
et al., EP 47005;
eidem, US 4382892 (1982, 1983 both to Daiichi). Total synthesis: H. Egawa
et al., Chem. Pharm. Bull. 34, 4098 (1986). Synthesis and activity of optical isomers: S. Atarashi
et al., ibid. 35, 1896 (1987). Antibacterial spectrum of racemate: K. Sato
et al., Antimicrob. Agents Chemother. 22, 548 (1982). Mechanism of differential activity of enantiomers: I. Morrissey
et al., ibid. 40, 1775 (1996). Toxicity data: H. Ohno
et al., Chemotherapy (Tokyo) 32, Suppl. 1, 1084 (1984). Pharmacology and clinical efficacy:
Infection 14, Suppl. 1, S1-S109 (1986). Symposium on pharmacokinetics and therapeutic use:
Scand. J. Infect. Dis. Suppl. 68, 1-69 (1990). Review of antibacterial spectrum, pharmacology, and clinical efficacy: J. P. Monk, D. M. Campoli-Richards,
Drugs 33, 346-391 (1987); of mechanism of action: K. Drlica,
Curr. Opin. Microbiol. 2, 504-508 (1999).
Properties: Colorless needles from ethanol, mp 250-257° (dec). LD50 in male, female mice, male, female rats (mg/kg): 5450, 5290, 3590, 3750 orally; 208, 233, 273, 276 i.v.; >10000, >10000, 7070, 9000 s.c. (Ohno).
Melting point: mp 250-257° (dec)
Toxicity data: LD50 in male, female mice, male, female rats (mg/kg): 5450, 5290, 3590, 3750 orally; 208, 233, 273, 276 i.v.; >10000, >10000, 7070, 9000 s.c. (Ohno)
Derivative Type: S-(-)-Form
CAS Registry Number: 100986-85-4; 138199-71-0 (hemihydrate)
Synonyms: Levofloxacin
Manufacturers' Codes: DR-3355
Trademarks: Cravit (Daiichi); Levaquin (Ortho-McNeil); Tavanic (Aventis); Quixin (Santen)
Literature References: Toxicity study: M. Kato
et al., Arzneim.-Forsch. 42, 365 (1992). Series of articles on pharmacology and toxicology:
ibid., 368-418. Clinical study in bacterial conjunctivitis: D. G. Hwang
et al., Br. J. Ophthalmol. 87, 1004 (2003).
Review: D. S. North
et al., Pharmacotherapy 18, 915-935 (1998).
Properties: Prepd as the hemihydrate; needles from ethanol + ethyl ether, mp 225-227° (dec). [a]D23 -76.9° (c = 0.385 in 0.5
N NaOH). Freely sol in glacial acetic acid, chloroform; sparingly sol in water. LD50 in male, female mice, male, female rats (mg/kg): 1881, 1803, 1478, 1507 orally (Kato).
Melting point: mp 225-227° (dec)
Optical Rotation: [a]D23 -76.9° (c = 0.385 in 0.5
N NaOH)
Toxicity data: Freely sol in glacial acetic acid, chloroform; sparingly sol in water. LD50 in male, female mice, male, female rats (mg/kg): 1881, 1803, 1478, 1507 orally (Kato)
Therap-Cat: Antibacterial.
Keywords: Antibacterial (Synthetic); Quinolones and Analogs.