Title: Lead Tetraacetate
CAS Registry Number: 546-67-8
Molecular Formula: C8H12O8Pb
Molecular Weight: 443.38
Percent Composition: C 21.67%, H 2.73%, O 28.87%, Pb 46.73%
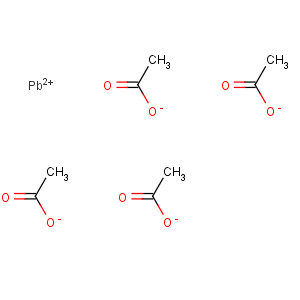
Line Formula: Pb(CH3COO)4
Literature References: Prepd from Pb3O4 and glacial acetic acid preferably in the presence of some acetic anhydride: Dimroth, Schweizer,
Ber. 56, 1375 (1923); Bailar,
Inorg. Synth. 1, 47 (1939); Baudler in
Handbook of Preparative Inorganic Chemistry vol. 1, G. Brauer, Ed. (Academic Press, New York, 2nd ed., 1963) p 767. Prepn by electrolysis: Fioshin, Gus'kov,
Dokl. Akad. Nauk SSSR 112, 303 (1957),
C.A. 51, 16146 (1957); Sataev
et al., Khim. Prom. (Moscow) 46, 892 (1970),
C.A. 74, 49005x (1971). Reviews of prepn and use as oxidizing agent: Criegee "Oxidations with Lead Tetraacetate" in
Oxidation in Organic Chemistry, Part A, K. B. Wiberg, Ed. (Academic Press, New York, 1965) pp 277-366; Zyka,
Pure Appl. Chem. 13, 569-581 (1966).
Properties: Colorless monoclinic prisms from glacial acetic acid. Turns pink easily. Unstable in air. Hydrolyzed by water with the formation of brown lead dioxide and acetic acid.
Avoid contact with skin. d417 2.228. mp 175-180°. Sol in hot glacial acetic acid, benzene, chloroform, tetrachloroethane, nitrobenzene. Dissolves in concd halogen acids with the formation of haloplumbic acids, H2PbX6. The dry material can be stored in sealed, evacuated ampuls.
Melting point: mp 175-180°
Density: d417 2.228
Use: Selective oxidizing agent in organic syntheses: Criegee,
Angew. Chem. 53, 321 (1940);
Newer Methods of Preparative Organic Chemistry (Interscience, N. Y., 1948) pp 1-17.