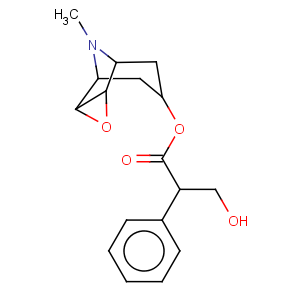
Title: Scopolamine
CAS Registry Number: 51-34-3
CAS Name: (a
S)-a-(Hydroxymethyl)benzeneacetic acid (1a,2b,4b,5a,7b)-9-methyl-3-oxa-9-azatricyclo[3.3.1.02,4]non-7-yl ester
Synonyms: 6b,7b-epoxy-1a
H,5a
H-tropan-3a-ol (-)-tropate; 6b,7b-epoxy-3a-tropanyl
S-(-)-tropate; 6,7-epoxytropine tropate; scopine tropate; tropic acid ester with scopine; hyoscine;
l-scopolamine
Trademarks: Scopoderm TTS (Novartis); Transcop (Recordati); Transderm Scop (Novartis)
Molecular Formula: C17H21NO4
Molecular Weight: 303.35
Percent Composition: C 67.31%, H 6.98%, N 4.62%, O 21.10%
Literature References: Anticholinergic, tropane alkaloid isolated from
Datura metel L.,
Scopola carniolica Jacq. and other
Solanaceae. Constituent of impure duboisine from
Duboisia myoporoides R. Br., pure duboisine is
l-hyoscyamine,
q.v. Isoln: A. Ladenburg,
Ann. 206, 274 (1881); E. Schmidt,
Arch. Pharm. 230, 207 (1892). Identity with hyoscine: O. Hesse,
Ann. 271, 100 (1892);
idem, J. Prakt. Chem. 66, 194 (1902). Absorption spectra: J. J. Dobbie, J. J. Fox,
J. Chem. Soc. 103, 1194 (1913). Resolution of isomers and review of early literature: H. King,
ibid. 1919, 476. Extraction procedure: F. Chemnitius,
J. Prakt. Chem. 120, 221 (1928). Structural studies: J. Gadamer, F. Hammer,
Arch. Pharm. 259, 122 (1921); K. Hess, O. Wahl,
Ber. 55, 1979 (1922); R. Willst?tter, E. Berner,
ibid. 56, 1079 (1923); W. Steffens,
Arch. Pharm. 262, 205 (1924). Configuration: G. Fodor,
Nature 170, 278 (1952); J. Meinwald,
J. Chem. Soc. 1953, 712. Review of stereochemistry: G. Fodor,
Tetrahedron 1, 86 (1957). Absolute configuration of tropic acid moiety: G. Fodor, G. Csepreghy,
Tetrahedron Lett. 1959, 16;
eidem, J. Chem. Soc. 1961, 3222. Synthesis of DL-form: G. Fodor
et al., Chem. Ind. (London) 1956, 764; P. Dobo
et al., J. Chem. Soc. 1959, 3461. Clinical evaluation in motion sickness: J. J. Brand, P. Whittingham,
Lancet 2, 232 (1970); in peripheral vertigo: T. Rahko, P. Karma,
J. Laryngol. Otol. 99, 653 (1985). Acute toxicity of the hydrobromide: Stockhaus, Wick,
Arch. Int. Pharmacodyn. Ther. 180, 155 (1969). Review of CNS effects in humans: D. J. Safer, R. P. Allen,
Biol. Psychiatry 3, 347-355 (1971); of use in anesthesia: L. E. Shutt, J. B. Bowles,
Anaesthesia 34, 476-490 (1979); of pharmacology and clinical efficacy: S. P. Clissold, R. C. Heel,
Drugs 29, 189-207 (1985). Comprehensive description: F. J. Muhtadi, M. M. A. Hassan,
Anal. Profiles Drug Subs. 19, 477-551 (1990).
Properties: Viscous liquid. pKa 7.55-7.81. [a]D20 -28° (c = 2.7). Sol in 9.5 parts water at 15°. Forms a cryst monohydrate, mp 59°. Freely sol in hot water, in alcohol, ether, chloroform, acetone. Sparingly sol in benzene, petr ether. Easily hydrolyzed by acids or alkalies. Dec on standing.
Melting point: mp 59°
pKa: pKa 7.55-7.81
Optical Rotation: [a]D20 -28° (c = 2.7)
Derivative Type: Hydrobromide trihydrate
CAS Registry Number: 114-49-8
Synonyms: Scopolammonium bromide
Molecular Formula: C17H21NO4.HBr.3H2O
Molecular Weight: 438.31
Percent Composition: C 46.58%, H 6.44%, N 3.20%, O 25.55%, Br 18.23%
Properties: Orthorhombic sphenoidal crystals from water, slightly efflorescent in dry air. mp 195° (after drying at 105° for 3 hours). [a]D25 -24 to -26° (c = 5, calculated on anhydrous basis). uv max (methanol) 246, 252, 258, 264 nm (A1%1cm 3.5, 4.0, 4.5, 3.0). pH of 0.05
M soln 5.85. One gram dissolves in 1.5 ml water, 20 ml alcohol. Slightly sol in chloroform. Practically insol in ether. LD50 in rats (mg/kg): 3800 s.c. (Stockhaus, Wick).
Melting point: mp 195° (after drying at 105° for 3 hours)
Optical Rotation: [a]D25 -24 to -26° (c = 5, calculated on anhydrous basis)
Absorption maximum: uv max (methanol) 246, 252, 258, 264 nm (A1%1cm 3.5, 4.0, 4.5, 3.0)
Toxicity data: LD50 in rats (mg/kg): 3800 s.c. (Stockhaus, Wick)
Derivative Type: Hydrochloride
CAS Registry Number: 55-16-3
Molecular Formula: C17H21NO4.HCl
Molecular Weight: 339.81
Percent Composition: C 60.09%, H 6.53%, N 4.12%, O 18.83%, Cl 10.43%
Properties: Crystals from acetone, mp 200°. Dihydrate, prisms from water, melts in water of crystn at 80°. Very sol in water and alcohol. pH of 0.05
M soln 5.85.
Melting point: mp 200°
Derivative Type: Methyl bromide
see Methscopolamine Bromide
Derivative Type: Methyl nitrate
CAS Registry Number: 6106-46-3
Synonyms: Methscopolamine nitrate
Molecular Formula: C17H21NO4.CH3NO3
Molecular Weight: 380.39
Percent Composition: C 56.83%, H 6.36%, N 7.36%, O 29.44%
Properties: Crystals. Freely sol in water, dil alcohol; slightly sol in abs alcohol.
Derivative Type: DL-Form
Synonyms: Atroscine
Properties: Dihydrate, chisel-shaped prisms from ethanol + water, mp 38-40°. Monohydrate, efflorescent crystals, mp 55-57°. Anhydrous, long prisms, mp 82-83°. Very slightly sol in water; sol in alc, chloroform, ether, oils.
Melting point: mp 38-40°; mp 55-57°; mp 82-83°
Therap-Cat: In treatment of motion sickness; antiemetic; antispasmodic; mydriatic; preanesthetic medicant.
Therap-Cat-Vet: Preanesthetic medicant.
Keywords: Antiemetic; Mydriatic; Antispasmodic; Antimuscarinic.