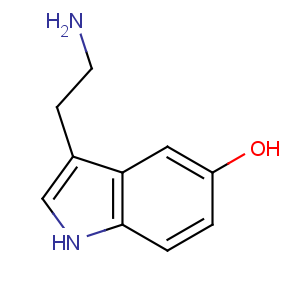
Title: Serotonin
CAS Registry Number: 50-67-9
CAS Name: 3-(2-Aminoethyl)-1
H-indol-5-ol
Synonyms: 5-hydroxytryptamine; 3-(b-aminoethyl)-5-hydroxyindole; 5-hydroxy-3-(b-aminoethyl)indole; enteramine; thrombocytin; thrombotonin; 5-HT
Molecular Formula: C10H12N2O
Molecular Weight: 176.22
Percent Composition: C 68.16%, H 6.86%, N 15.90%, O 9.08%
Literature References: Vasoactive amine found in tissues and fluids of vertebrates and invertebrates. Extraction of enteramine from rabbit tissue, pharmacology: V. Erspamer,
Arch. Exp. Pathol. Pharmakol. 196, 343 (1940). Isoln of 5-HT from beef serum: M. M. Rapport
et al., Science 108, 329 (1948);
eidem, J. Biol. Chem. 176, 1237, 1243 (1948); M. M. Rapport,
ibid. 180, 961 (1949). Identity with enteramine, the enterochromaffin cell hormone: V. Erspamer, B. Asero,
Nature 169, 800 (1952). Ultrastructural localization in enterochromaffin cells: R. D. Dey, J. Hoffpauir,
J. Histochem. Cytochem. 32, 661 (1984). Synthesis starting with 5-benzyloxyindole: M. E. Speeter
et al., J. Am. Chem. Soc. 73, 5514 (1951); K. E. Hamlin,
US 2715129 (1955 to Abbott). Alternate routes: R. Justoni, R. Pessina,
US 2947757 (1960 to Vismara). Pharmacology: G. Reid, M. Rand,
Nature 169, 801 (1952). Existence of multiple serotonin receptors: S. J. Peroutka, S. H. Snyder,
Mol. Pharmacol. 16, 687 (1979); G. Engel
et al., Arch. Pharmacol. 324, 116 (1983); W. Feniuk
et al., Eur. J. Pharmacol. 96, 71 (1983). Extraneural synthesis in CNS: C. Maruki
et al., J. Neurochem. 43, 316 (1984). Effect on smooth muscle: W. Feniuk
et al., loc. cit. Activity as neurotransmitter: S. G. Griffith
et al., Brain Res. 247, 388 (1982). Role in hemostasis: F. De Clerck
et al., Agents Actions 15, 627 (1984). Series of articles on role in hypertension, Raynaud's phenomenon and platelet activation:
J. Cardiovasc. Pharmacol. 7, Suppl. 7, 1-182 (1985). Review of role in migraine: E. T. MacKenzie
et al., Cephalalgia 5, 69-78 (1985).
Review: V. Erspamer in E. Jucker,
Prog. Drug Res. 3, 151-367 (1961). Books:
Serotonin in Health and Disease vols. I-V, W. B. Essman, Ed. (Spectrum, New York, 1978);
Adv. Exp. Med. 133, entitled "Serotonin: Current Aspects of Neurochemistry and Function," B. Haber, Ed. (1981) 824 pp;
Serotonin and the Cardiovascular System, P. M. Vanhoutte, Ed. (Raven Press, New York, 1985) 280 pp.
See also Bufotenine and Ketanserin.
Derivative Type: Hydrochloride
Molecular Formula: C10H12N2O.HCl
Molecular Weight: 212.68
Percent Composition: C 56.47%, H 6.16%, N 13.17%, O 7.52%, Cl 16.67%
Properties: Hygroscopic crystals, sensitive to light, mp 167-168°. Water sol. Aq solns are stable at pH 2-6.4.
Melting point: mp 167-168°
Derivative Type: Complex with creatinine sulfate, monohydrate
Trademarks: Antemovis (Vister)
Molecular Formula: C14H21N5O6S.H2O
Molecular Weight: 405.43
Percent Composition: C 41.47%, H 5.72%, N 17.27%, O 27.62%, S 7.91%
Properties: Plates, 215° dec. uv max (water at pH 3.5): 275 nm (e 15000). pK1¢ = 4.9; pK2¢ = 9.8. pH of 0.01 molar aq soln: 3.6. Sol in glacial acetic acid. Very sparingly sol in methanol, 95% ethanol. Insol in abs ethanol, acetone, pyridine, chloroform, ethyl acetate, ether, benzene.
pKa: pK1¢ = 4.9; pK2¢ = 9.8
Absorption maximum: uv max (water at pH 3.5): 275 nm (e 15000)