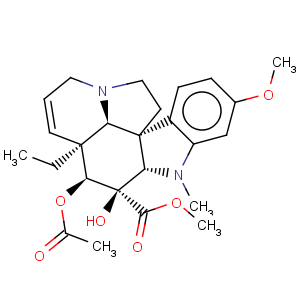
Title: Vindoline
CAS Registry Number: 2182-14-1
CAS Name: (2b,3b,4b,5a,12b,19a)-4-(Acetyloxy)-6,7-didehydro-3-hydroxy-16-methoxy-1-methylaspidospermidine-3-carboxylic acid methyl ester
Molecular Formula: C25H32N2O6
Molecular Weight: 456.53
Percent Composition: C 65.77%, H 7.07%, N 6.14%, O 21.03%
Literature References: Major alkaloid from the leaves of
Vinca rosea Linn. (
Catharanthus roseus G. Don.),
Apocynaceae; occurs naturally as the (-)-form: Gorman
et al., J. Am. Pharm. Assoc. 48, 256 (1959); Svoboda
et al., ibid. 659; Moza, Trojánek,
Collect. Czech. Chem. Commun. 28, 1419 (1963). Structure: Gorman
et al., J. Am. Chem. Soc. 84, 1058 (1962); Neuss,
Bull. Soc. Chim. Fr. 1963, 1509. Stereochemistry: Moncrief, Lipscomb,
J. Am. Chem. Soc. 87, 4963 (1965). Review of chemistry: Neuss
et al., Adv. Chemother. 1, 133 (1964). Total synthesis of (±)-vindoline: Ando
et al., J. Am. Chem. Soc. 97, 6880 (1975); Y. Ban
et al., Tetrahedron Lett. 1978, 151; J. P. Kutney
et al., J. Am. Chem. Soc. 100, 4220 (1978). Lacks physiological activity alone but is contained as the pentacyclic moiety in the antineoplastic agents vinblastine and vincristine,
q.q.v.
Properties: Needles from acetone + petr ether, mp 164-165°; prisms, mp 174-175°; [a]D20 -18° (chloroform); pKa 5.5 in 66% DMF (Moza, Trojánek). Also reported as crystals, mp 154-155°; [a]D27 +42° (chloroform) (Gorman). uv max (ethanol): 212, 250, 304 nm (log e 4.49, 3.74, 3.57).
Melting point: mp 164-165°; mp 174-175°; mp 154-155°
pKa: pKa 5.5 in 66% DMF (Moza, Trojánek)
Optical Rotation: [a]D20 -18° (chloroform); [a]D27 +42° (chloroform)
Absorption maximum: uv max (ethanol): 212, 250, 304 nm (log e 4.49, 3.74, 3.57)
Derivative Type: Hydrochloride
Molecular Formula: C25H32N2O6.HCl
Molecular Weight: 492.99
Percent Composition: C 60.91%, H 6.75%, N 5.68%, O 19.47%, Cl 7.19%
Properties: Crystals from acetone, mp 161-164°.
Melting point: mp 161-164°
Derivative Type: Demethoxyvindoline
CAS Registry Number: 5231-60-7
Synonyms: Vindorosine; vindolidine
Molecular Formula: C24H30N2O5
Molecular Weight: 426.51
Percent Composition: C 67.59%, H 7.09%, N 6.57%, O 18.76%
Literature References: Structure: Moza, Trojánek,
Collect. Czech. Chem. Commun. 28, 1427 (1963).
Properties: Needles from benzene + petr ether, mp 167°. [a]D16 -31° (chloroform). uv max (methanol): 250, 302 nm (log e 3.98, 3.52).
Melting point: mp 167°
Optical Rotation: [a]D16 -31° (chloroform)
Absorption maximum: uv max (methanol): 250, 302 nm (log e 3.98, 3.52)