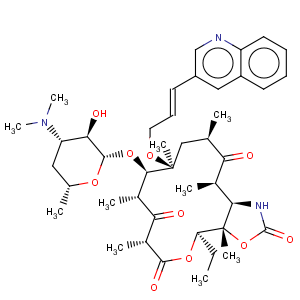
Title: Cethromycin
CAS Registry Number: 205110-48-1
CAS Name: (3a
S,4
R,7
R,9
R,10
R,11
R,13
R,15
R,15a
R)-4-Ethyloctahydro-3a,7,9,11,13,15-hexamethyl-11-[[3-(3-quinolinyl)-2-propenyl]oxy]-10-[[3,4,6-trideoxy-3-(dimethylamino)-b-D-xylohexopyranosyl]oxy]-2
H-oxacyclotetradecino[4,3-
d]oxazole-2,6,8,14(1
H,7
H,9
H)-tetrone
Manufacturers' Codes: A-195773; ABT-773
Molecular Formula: C42H59N3O10
Molecular Weight: 765.93
Percent Composition: C 65.86%, H 7.76%, N 5.49%, O 20.89%
Literature References: Semisynthetic macrolide antibiotic of the ketolide class; structurally similar to telithromycin,
q.v. Prepn: Y. S. Or
et al., WO 9809978;
eidem, US 5866549 (1998, 1999 both to Abbott);
eidem, J. Med. Chem. 43, 1045 (2000). Improved prepn: D. J. Plata
et al., Tetrahedron 60, 10171 (2004). Comparative
in vitro antibacterial spectrum: A. M. Nilius
et al., Antimicrob. Agents Chemother. 45, 2163 (2001). Mechanism of action study: W. S. Champney, J. Pelt,
Curr. Microbiol. 45, 155 (2002). HPLC-MS determn in biological samples: Q. Ren
et al., J. Chromatogr. Sci. 41, 494 (2003). Clinical pharmacokinetics: M. W. Pletz
et al., Antimicrob. Agents Chemother. 47, 1129 (2003). Reviews of development and therapeutic potential: L. E. Lawrence,
Curr. Opin. Invest. Drugs 2, 766-772 (2001); T. J. Dougherty, J. F. Barrett,
Expert Opin. Invest. Drugs 10, 343-351 (2001).
Properties: White crystalline solid, mp 211-213°.
Melting point: mp 211-213°
Therap-Cat: Antibacterial.
Keywords: Antibacterial (Antibiotics); Macrolides.