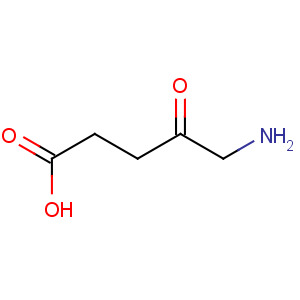
Title: d-Aminolevulinic Acid
CAS Registry Number: 106-60-5
CAS Name: 5-Amino-4-oxopentanoic acid
Synonyms: ALA
Molecular Formula: C5H9NO3
Molecular Weight: 131.13
Percent Composition: C 45.80%, H 6.92%, N 10.68%, O 36.60%
Line Formula: H2NCH2COCH2CH2COOH
Literature References: Naturally occurring amino acid; precursor of tetrapyrroles in the biosynthesis of chlorophyll and heme. Prepn: R. W. Wynn, A. H. Corwin,
J. Org. Chem. 15, 203 (1950); A. A. Marei, R. H. Raphael,
J. Chem. Soc. 1958, 2624; C. Herdeis, A. Dimmerlung,
Arch. Pharm. 317, 304 (1984). Role in tetrapyrrole biosynthesis: D. Shemin, C. S. Russell,
J. Am. Chem. Soc. 75, 4873 (1953). Enhancement of chlorophyll formation: E. C. Sisler, W. H. Klein,
Physiol. Plant. 16, 315 (1963). HPLC determn in plasma: K. Miyajima
et al., J. Chromatogr. B 654, 165 (1994). Review of role in chlorophyll biosynthesis and potential as photodynamic herbicide: C. A. Rebeiz
et al., Enzyme Microb. Technol. 6, 390-401 (1984). Review of role in heme biosynthesis and clinical experience in photodynamic therapy: Q. Peng
et al., Cancer 79, 2282-2308 (1997).
Derivative Type: Hydrochloride
CAS Registry Number: 5451-09-2
Trademarks: Levulan (Dusa)
Molecular Formula: C5H10ClNO3
Molecular Weight: 167.59
Percent Composition: C 35.83%, H 6.01%, Cl 21.15%, N 8.36%, O 28.64%
Properties: Needles from methanol-ether, mp 144-147° (dec).
Melting point: mp 144-147° (dec)
Therap-Cat: Antineoplastic (photosensitizer).
Keywords: Antineoplastic (Photosensitizer).