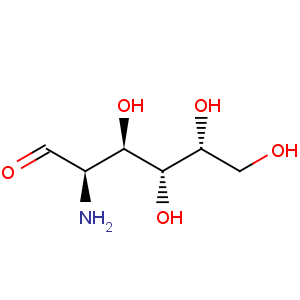
Title: D-Galactosamine
CAS Registry Number: 7535-00-4
CAS Name: 2-Amino-2-deoxy-D-galactose
Synonyms: chondrosamine; GalN
Molecular Formula: C6H13NO5
Molecular Weight: 179.17
Percent Composition: C 40.22%, H 7.31%, N 7.82%, O 44.65%
Literature References: Amino sugar isolated from chondroitin sulfate,
q.v.: P. A. Levene, F. B. La Forge,
J. Biol. Chem. 18, 123 (1914). Sepn of a- and b-anomers: P. A. Levene,
ibid. 57, 337 (1923). Synthesis: S. P. James
et al., Nature 156, 308 (1945)
eidem, J. Chem. Soc. 1946, 625; R. Kuhn, W. Kirschenlohr,
Ann. 600, 126 (1956); P. A. Gent
et al., J. Chem. Soc. Perkin Trans. 1 1972, 277. Chemistry: D. Horton in
The Amino Sugars Vol. 1A, R. W. Jeanloz, Ed. (Academic, New York, 1969) pp 133-145. Inducer of exptl hepatitis: D. Keppler
et al., Exp. Mol. Pathol. 9, 279 (1968); K. Decker, D. Keppler in
Progress in Liver Diseases Vol. IV, H. Popper, F. Schaffner, Eds. (Grune & Stratton, New York, 1972) p 183. Powerful inhibitor of hepatic RNA synthesis: D. Keppler
et al., J. Biol. Chem. 249, 211 (1974); T. Anukarahanonta
et al., Eur. J. Cancer 16, 1171 (1980).
Derivative Type: Hydrochloride
Molecular Formula: C6H14ClNO5
Molecular Weight: 215.63
Percent Composition: C 33.42%, H 6.54%, Cl 16.44%, N 6.50%, O 37.10%
Properties: Crystals, mp 180° (dec). Shows mutarotation. a-Form: [a]D23 +124° ? +93° (water). b-Form: [a]D23 +47° ? +93° (water).
Melting point: mp 180° (dec)
Optical Rotation: a-Form: [a]D23 +124° ? +93° (water); b-Form: [a]D23 +47° ? +93° (water)