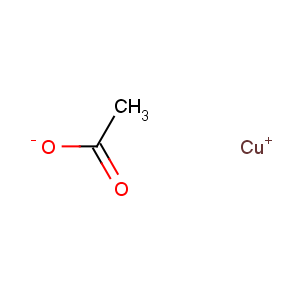
Title: Cuprous Acetate
CAS Registry Number: 598-54-9
CAS Name: Acetic acid copper(1+) salt
Molecular Formula: C2H3CuO2
Molecular Weight: 122.59
Percent Composition: C 19.59%, H 2.47%, Cu 51.84%, O 26.10%
Line Formula: CH3COOCu
Literature References: Obtained as a sublimate by heating cupric acetate
in vacuo to temps above 220°: Angel, Harcourt,
J. Chem. Soc. 81, 1385 (1902); prepn from cuprous oxide and acetic acid-acetic anhydride: Shimizu, Weller,
J. Am. Chem. Soc. 74, 4469 (1952). Crystal structure: T. Ogura
et al., J. Am. Chem. Soc. 95, 949 (1973).
Properties: Transparent, leafy crystals. Volatilizes on heating; dec on strong heating. Rapidly hydrolyzed by water with the formation of yellow Cu2O.