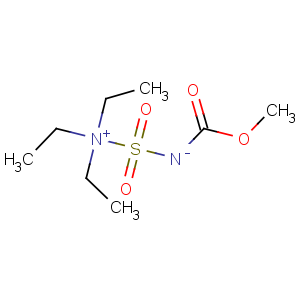
Title: Burgess Reagent
CAS Registry Number: 29684-56-8
CAS Name: N,
N-Diethyl-
N-[[(methoxycarbonyl)amino]sulfonyl]ethanaminium inner salt
Synonyms: (methoxycarbonylsulfamoyl)triethylammonium hydroxide inner salt; methyl(carboxysulfamoyl)triethylammonium hydroxide inner salt; methyl-
N-(triethylammoniumsulfonyl)carbamate
Molecular Formula: C8H18N2O4S
Molecular Weight: 238.30
Percent Composition: C 40.32%, H 7.61%, N 11.76%, O 26.86%, S 13.46%
Literature References: Dehydrating agent. Synthesis method: G. M. Atkins, Jr., E. M. Burgess,
J. Am. Chem. Soc. 90, 4744 (1968). Prepn and applications in dehydration reactions: E. M. Burgess
et al.,
ibid.
92, 5224 (1970);
eidem,
J. Org. Chem. 38, 26 (1973). Prepn and reaction with primary alcohols to form urethanes: E. M. Burgess
et al., Org. Synth. 56, 40 (1977). Reaction with epoxides: U. Rinner
et al.,
Synlett 2003, 1247. Review of chemistry: C. Lamberth,
J. Prakt. Chem. Chem.-Ztg. 342, 518-522 (2000); K. C. Nicolaou
et al.,
Chem. Eur. J. 10, 5581-5606 (2004).
Properties: Colorless needles from toluene, mp 71-72°.
Melting point: mp 71-72°
Use: Reagent for dehydration of secondary and tertiary alcohols; source of heteroatoms (N, O, S) in organic synthesis.