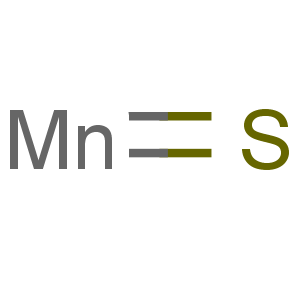Title: Manganese Sulfide
CAS Registry Number: 18820-29-6
Synonyms: Manganese monosulfide
Molecular Formula: MnS
Molecular Weight: 87.00
Percent Composition: Mn 63.15%, S 36.86%
Literature References: Occurs in nature as the mineral
alabandite or
manganblende. Prepn: Mehmed, Haraldsen,
Z. Anorg. Chem. 235, 194 (1938).
Properties: Pink, green or brown-green powder. Three cryst modifications: a-form, green cubic crystals; b-form, red cubic crystals; g-form, red hexagonal crystals. Practically insol in water; sol in dil acids. In moist condition it readily oxidizes in air to the sulfate.