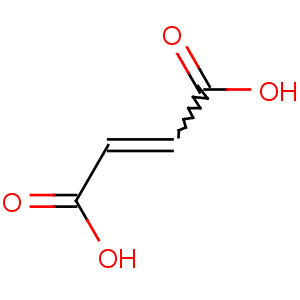
Title: Fumaric Acid
CAS Registry Number: 110-17-8
CAS Name: (2
E)-2-Butenedioic acid
Synonyms: trans-1,2-ethylenedicarboxylic acid; allomaleic acid; boletic acid
Molecular Formula: C4H4O4
Molecular Weight: 116.07
Percent Composition: C 41.39%, H 3.47%, O 55.14%
Literature References: Occurs in many plants,
e.g., in
Fumaria officinalis L.,
Fumariaceae, in
Boletus scaber Bull.,
Boletaceae, and in
Fomes igniarius (Fries) Kickx.,
Polyporaceae. Essential to vegetable and animal tissue respiration. Prepd industrially from glucose by the action of fungi such as
Rhizopus nigricans: Foster, Waksman,
J. Am. Chem. Soc. 61, 127 (1939). Laboratory prepn by the oxidation of furfural with sodium chlorate in the presence of vanadium pentoxide: Milas,
Org. Synth. coll. vol. II, 302 (1943). Molecular structure: J. L. Derissen,
J. Mol. Struct. 38, 177 (1977).
Review: W. D. Robinson, R. A. Mount in
Kirk-Othmer Encyclopedia of Chemical Technology vol. 14 (Wiley-Interscience, New York, 3rd ed., 1981) pp 770-793.
Properties: Monoclinic, prismatic needles or leaflets from water. d 1.625. Sublimes at 200°. Sublimes at 165° at 1.7 mm pressure. Partial carbonization and formation of maleic anhydride occur at 230° (open vessel). mp 287° (closed capillary, rapid heating). pK1 (25°): 3.03; pK2: 4.54. Absorption spectrum: Macbeth, Stewart,
J. Chem. Soc. 111, 830 (1917). Soly in 100 g water at 25°: 0.63 g; at 40°: 1.07 g; at 60°: 2.4 g; at 100°: 9.8 g; in 100 g 95% alcohol at 30°: 5.76 g; in 100 g acetone at 30°: 1.72 g; in 100 g ether at 25°: 0.72 g. Almost insol in olive oil, chloroform, carbon tetrachloride, benzene, xylene, molten camphor, liq ammonia.
Melting point: mp 287° (closed capillary, rapid heating)
pKa: pK1 (25°): 3.03; pK2: 4.54
Density: d 1.625
Derivative Type: Monomethyl ester
Molecular Formula: C5H6O4
Molecular Weight: 130.10
Percent Composition: C 46.16%, H 4.65%, O 49.19%
Properties: Prisms from alc, mp 144.5°.
Melting point: mp 144.5°
Derivative Type: Dimethyl ester
Molecular Formula: C6H8O4
Molecular Weight: 144.13
Percent Composition: C 50.00%, H 5.59%, O 44.40%
Properties: Crystals, mp 102°. bp 192°.
Melting point: mp 102°
Boiling point: bp 192°
Use: Substitute for tartaric acid in beverages and baking powders; as a replacement or partial replacement for citric acid in fruit drinks. As an antioxidant. Manuf polyhydric alcohols, synthetic resins. As mordant in dyeing.